
Nulogy vs. Trustwell
Both FSMA 204-ready. Only one does everything else.
Trustwell built a traceability platform and added compliance around it with FoodLogiQ. Nulogy built a unified FSQA platform – and FSMA 204 traceability is part of it.
Same traceability capability. Plus audits, QMS, EHS, supplier qualification, corrective action, and production monitoring. One platform, one data model, no gaps.
SOC 2 Type II
iOS & Android
Built for FSQA
Trusted by the World’s Leading Manufacturers











END-TO-END Quality & Compliance Management
Nulogy vs. Trustwell
When it comes to FSMA 204, Nulogy and Trustwell are both ready. The difference is what surrounds it. Trustwell’s platform is built around ingredient traceability — everything else is an add-on or an integration.
Nulogy is built around your operation: plants, lines, suppliers, people, and products. FSMA 204 compliance lives inside a full QMS, EHS, and supplier audit platform — not alongside it.
Straightforward Pricing. No Surprises.
Nulogy is priced by facility — so your costs scale predictably as you grow, without getting nickel-and-dimed for add-ons. Whether you’re running one site or a hundred, you’ll know exactly what you’re paying for.
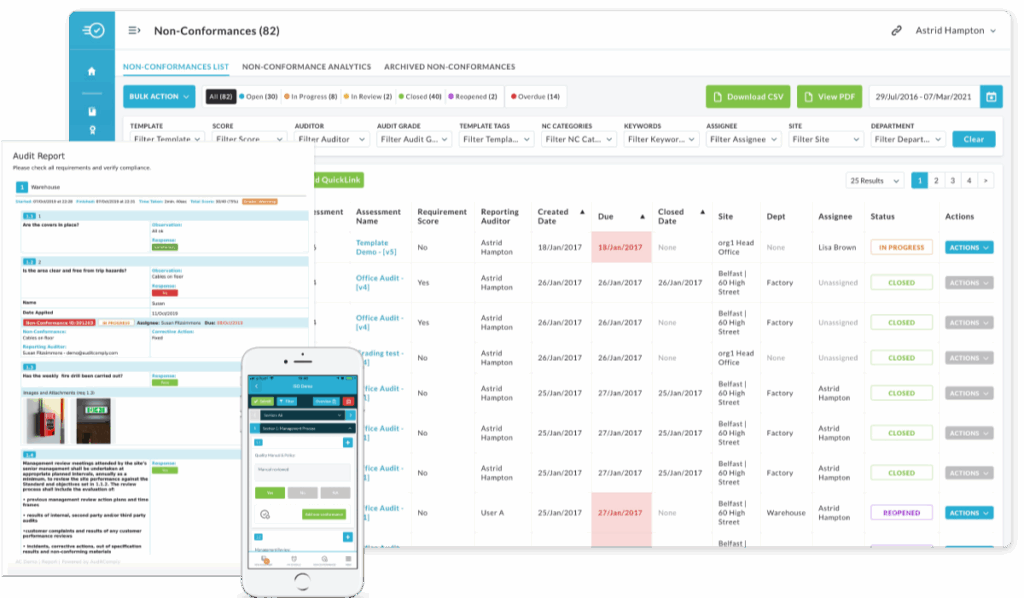
FSMA 204-ready. And the platform your auditors prefer.
Both Nulogy and Trustwell meet FSMA 204 requirements — CTEs, lot-level tracking, one-up/one-down traceability. But your FDA auditor isn’t only asking about traceability records. They’re asking about preventive controls, supplier verification, hazard analysis, and corrective actions. Nulogy covers all of it in one system. Trustwell covers one pillar.
- Full traceability, built in
- Not a standalone compliance tool
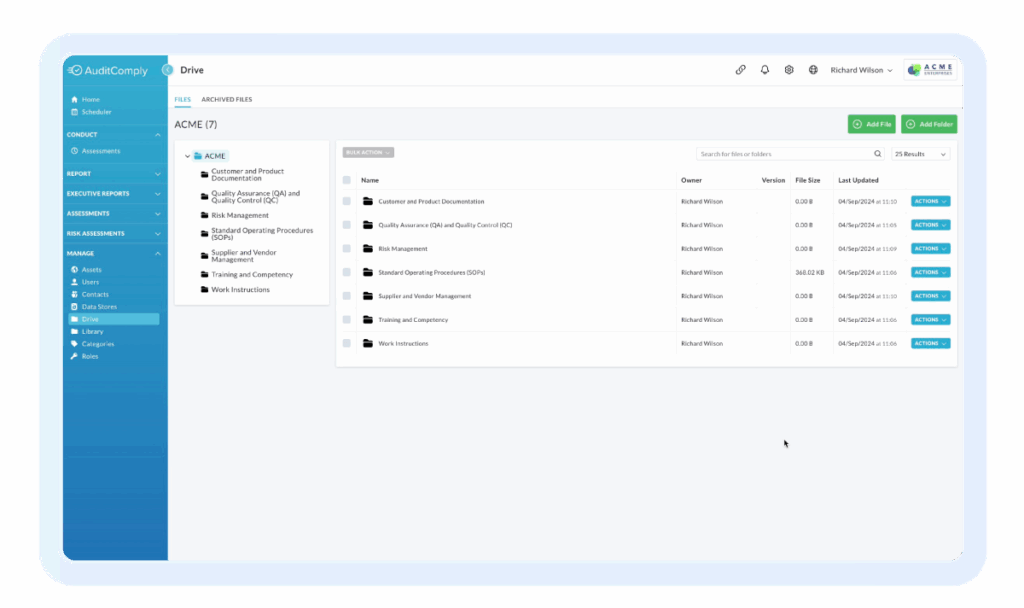
FoodLogiQ customers still need a separate audit platform. Nulogy is it.
Trustwell has no meaningful audit capability — no supplier audits, no internal quality audits, no CAPA workflows. Most Trustwell customers end up managing audits in spreadsheets or buying a second tool. Nulogy handles supplier qualification, internal audits, corrective actions, and compliance trending in one platform.
- Built-in audit management
- No separate audit tools
One platform. Not a traceability tool plus a spreadsheet.
Most Trustwell FoodLogiQ customers still manage plant audits, EHS incidents, and internal quality workflows in separate tools. Nulogy replaces all of it — QMS, EHS, supplier audits, CAPA, document control, and production monitoring in one configurable platform. One data model. One source of truth across every site.
- QMS, EHS, audits, production
- One platform, no gaps
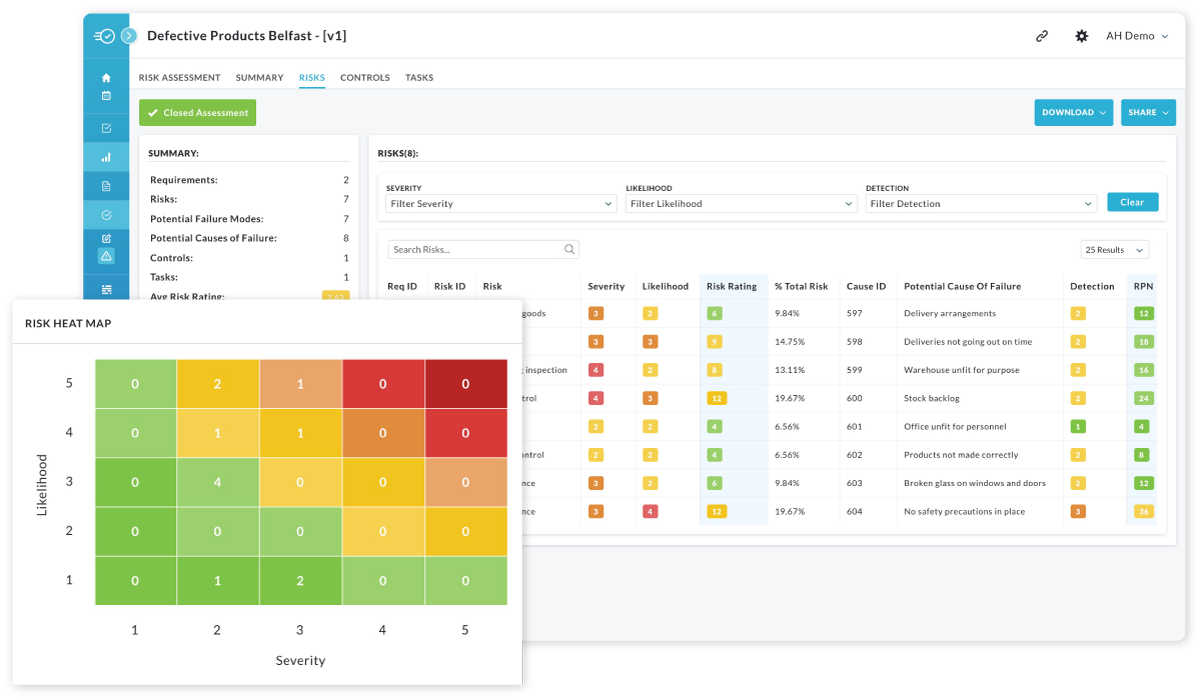

Sysco Chose Nulogy to Unify Quality Operations Across 180+ Sites
Sysco — one of the world’s largest food distributors — needed a quality platform that could handle multi-site complexity, custom audit workflows, and real-time visibility at enterprise scale. They chose Nulogy.
Today, Nulogy runs quality and FSQA operations across 180+ Sysco sites — with a single configurable platform that scales from one operator’s entry to a corporate KPI.
"We required a solution that was mobile, automated our data and could be accessed in real time to ensure we are meeting quality standards across the supply chain. Nulogy delivered."
Jonny Elgin
QA Manager, Sysco








END-TO-END FOOD QUALITY Management SYSTEM
Unite Suppliers, People, Quality & Production to Improve Plant Performance & Visibility
Empowering food & beverage teams with technology to track suppliers, drive a culture of safety, boost quality and increase yield.





























