FSMA Module
FSMA Food Traceability Software
Frameworks
FSMA Food Traceability Software
Track Your Products Every Step of the Way.
From Farm to Fork
FDA Inspection Readiness
End-to-End Traceability
Faster, More Accurate Recalls
Stay ready and FSMA 204 compliant with a system built for real-time food safety and traceability
Nulogy Quality & Compliance is your only purpose-built solution for delivering real-time food traceability and 24-hour responsiveness to quality requests. Track every step of the manufacturing lifecycle with ease, including Critical Tracking Events (CTEs), Key Data Elements (KDEs), and Traceability Lot Codes (TLCs), all in one centralized platform.
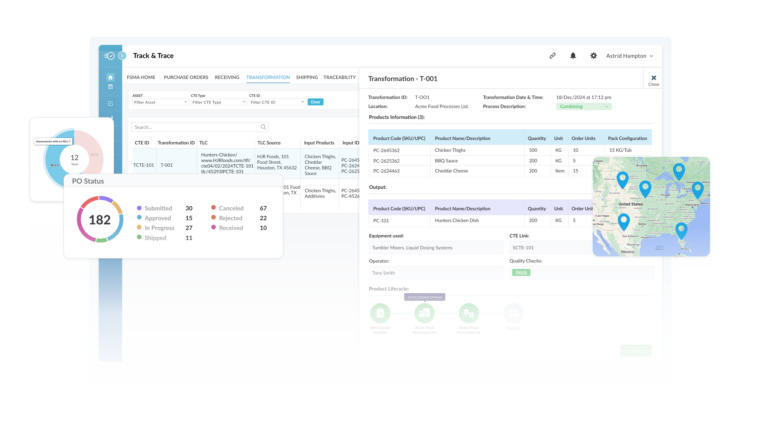
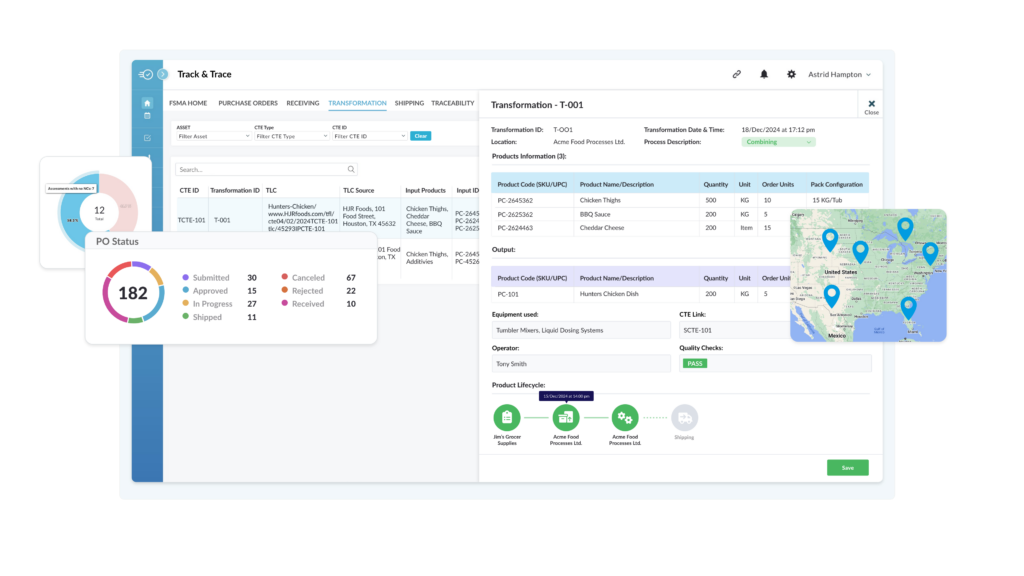
Purpose-Built CTE Register
Track all CTE events, and search and report instantly against a CTE record, TLC, product code, supplier or customer. Track progress and activity at a glance using detailed filters.
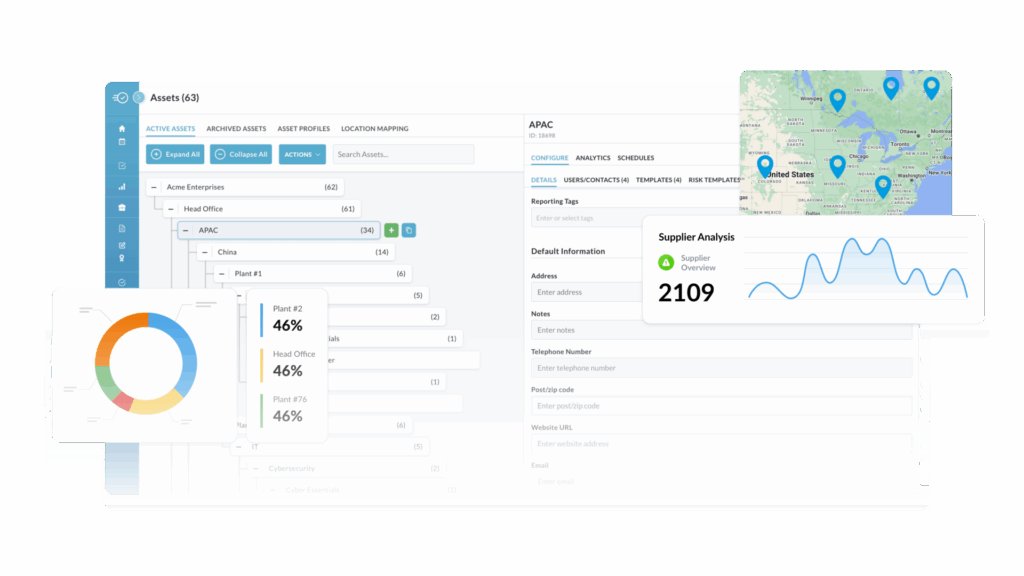
Supply Chain Traceability
Easily track your food supply chain end-to-end, including CTEs, KDEs, and TLCs, from raw material sourcing to final product delivery.
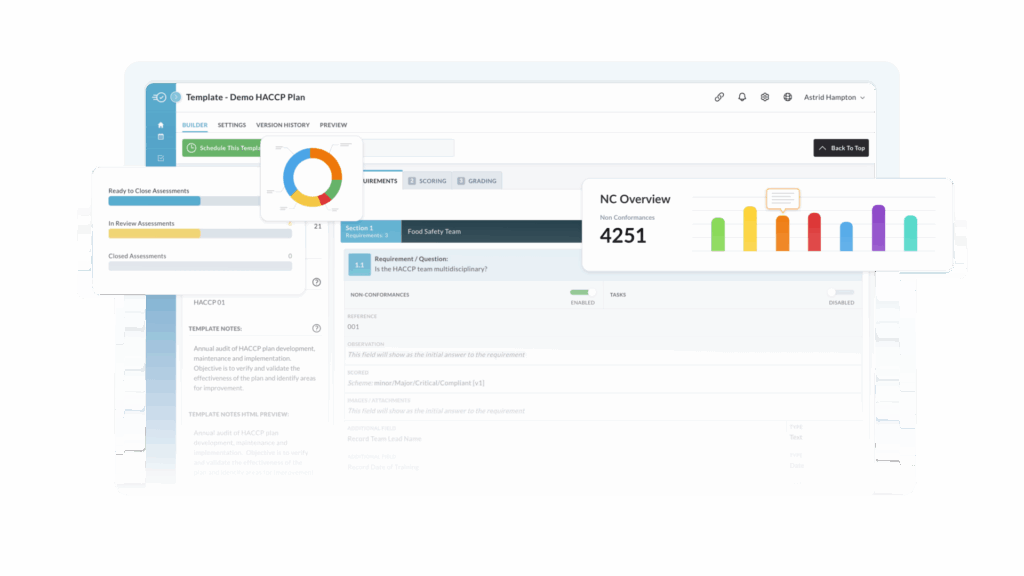
Automate FDA Reporting
Retrieve records in 24 hours, instantly identify contamination sources and react faster with real-time alerts to food safety risks.
SOME OF NULOGY’S CLIENTS
Trusted by Leading Food Manufacturers Worldwide





Enhance traceability to prevent recalls and achieve comprehensive visibility from source to customer
Nulogy’s platform streamlines compliance and enhances food safety and traceability.
KDE & CTE Capture
Automatically records required traceability data at every key supply chain event to ensure compliance with the FSMA 204 Rule.
End-to-End Lot Genealogy
Connects ingredients to finished products, enabling full traceability forward and backward across the supply chain.
Real-Time Data Capture
Uses scanning and automation to capture data instantly, reducing errors and ensuring fast access during audits or recalls.
Unique Identification & Labelling
Assigns standardized lot and product identifiers to maintain consistent, accurate tracking across systems and partners.
Recall Readiness & Mock Recalls
Allows rapid identification and simulation of affected products to execute precise and efficient recalls.
Integrated Systems (MES/WMS/QMS)
Connects traceability with manufacturing, warehousing, and quality systems to eliminate data silos and gaps.
Audit Trails & Data Integrity
Logs all data changes with timestamps and user tracking to ensure accuracy and inspection readiness.
Supply Chain Data Exchange & FDA Reporting
Enables seamless sharing of traceability data with partners and regulators like the FDA.
Automated Reporting & Fast Response
Generates compliant traceability reports quickly to meet regulatory deadlines and audit requests.

FSMA 204: Compliance Made Easy with Nulogy
Explore the key requirements of FSMA Section 204, tackle compliance challenges, and learn practical strategies for implementing advanced traceability systems to ensure regulatory success.
Frequently Asked Questions About FSMA 204 Compliance
Learn more about Nulogy’s traceability modules can easily enable FSMA 204 compliance.
What is FSMA 204, and why is it important?
FSMA 204 is a rule under the U.S. Food Safety Modernization Act that requires companies to maintain detailed traceability records for certain high-risk foods. Its goal is to enable rapid identification and removal of contaminated food from the supply chain, reducing the scale and impact of foodborne illness outbreaks.
What are the requirements to become FSMA 204 compliant?
To become compliant with FSMA 204, companies handling certain foods must implement end-to-end traceability across the supply chain, including:
Maintaining a written traceability plan
Capturing Key Data Elements (KDEs)
Recording Critical Tracking Events (CTEs)
Assigning traceability lot codes
Retaining and quickly sharing records with regulators.
When do food manufacturers need to be FSMA 204 compliant?
Food manufacturers and other covered companies must be fully compliant with FSMA 204 by July 21, 2028. By that date, organizations that manufacture, process, pack, or hold foods on the Food Traceability List must have the required traceability systems, records, and processes in place.
Who is responsible for ensuring FSMA 204 compliance for a food company?
Responsibility for FSMA 204 compliance ultimately lies with the food manufacturer, but operational accountability is typically shared across several internal roles. Most organizations assign day-to-day oversight to Food Safety, Quality Assurance (FSQA), or regulatory compliance teams, while leadership remains legally responsible for ensuring the company meets the rule’s requirements.
What high-risk foods are subject to increased data requirements?
FSMA 204 focuses on foods that have historically posed higher food safety risks—particularly fresh produce, seafood, soft cheeses, eggs, nut butters, and ready-to-eat deli foods—and requires detailed traceability data for them across the supply chain.
Nulogy's Track, Trace & Recall Platform
Helping you ensuring compliance, reducing risk & elevating food safety standards
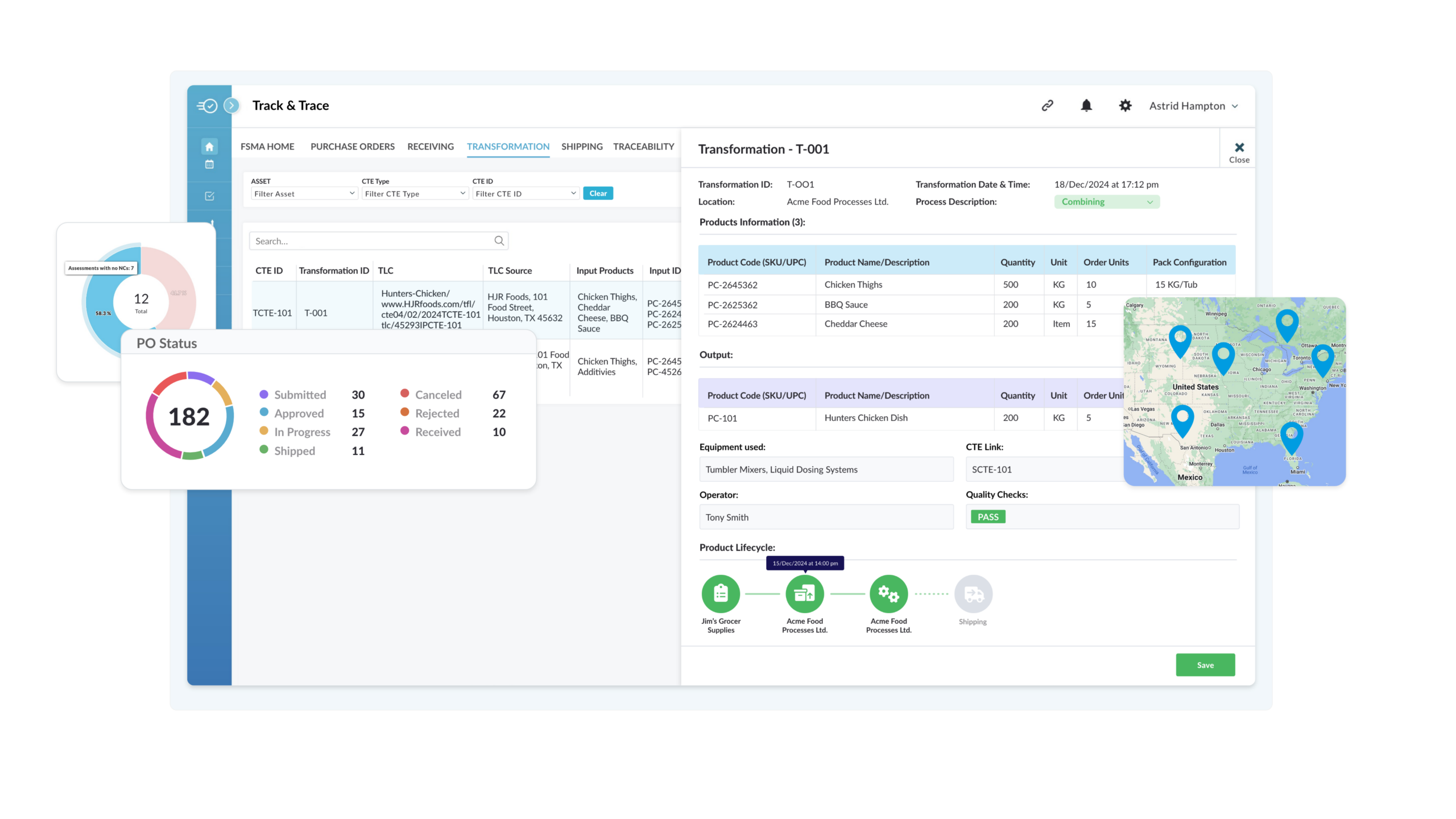
Get a Full Supply Chain View and Dive into CTEs
From field to production floor, Nulogy Traceability ensures your teams have instant access to critical data for streamlined compliance and enhanced operational efficiency.
- See every product movement from origin to customer
- Gain precise visibility into when, where, and how products change
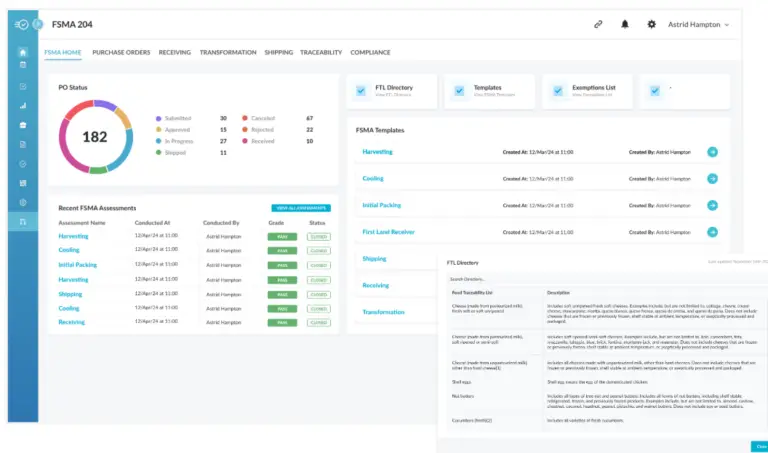
Trace Ingredients from Source to Consumer
Gain full visibility into your product’s ingredient journey with Nulogy’s traceability software, enabling seamless tracking at every level.
- Monitor every ingredient from origin to end product
- Ensure transparency with end-to-end visibility
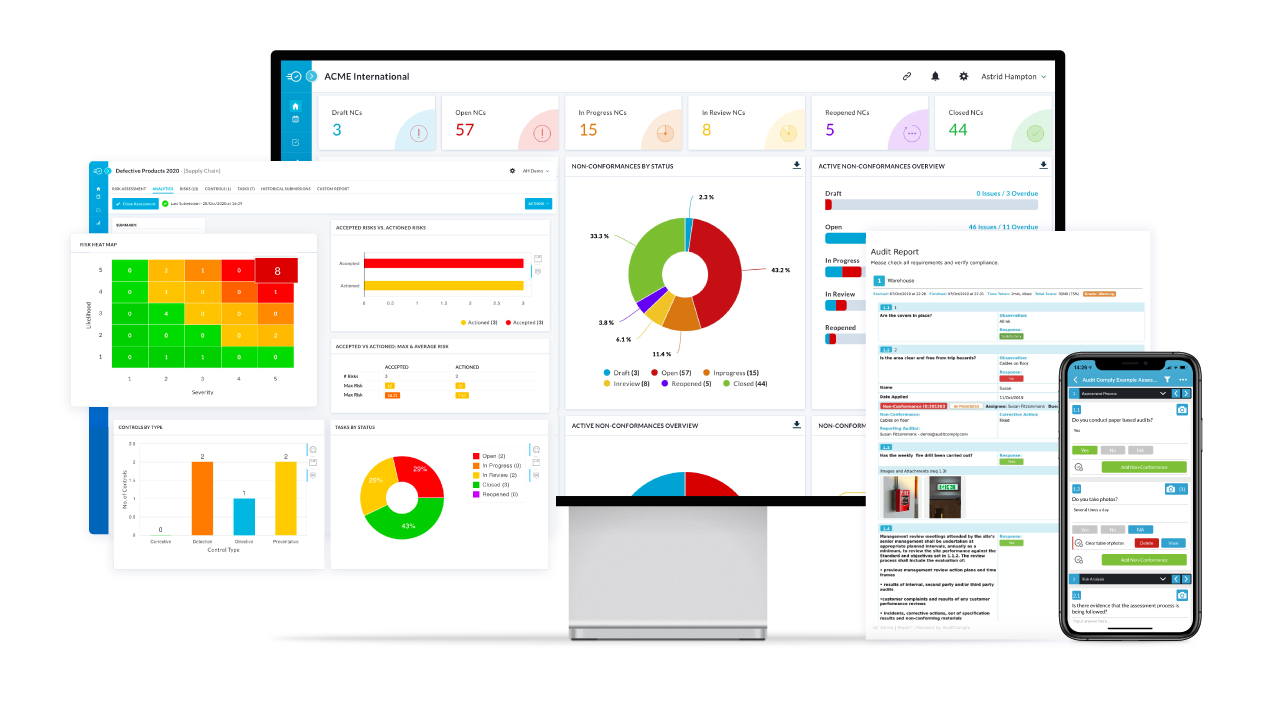
Ensure 24-Hour FDA Reporting with Lot-Level Traceability
Nulogy’s advanced food traceability software helps you swiftly identify, manage, and resolve food adulteration incidents, meeting FDA reporting demands with speed and precision.
- Instantly retrieve and share traceability records
- Pinpoint issues quickly to minimize risk

Interested in learning more?
Read more customer success stories or check out other frameworks.

